
SYDNEY, April 21, 2022 /PRNewswire/ -- Clarity Pharmaceuticals (ASX: CU6) ("Clarity"), a clinical-stage radiopharmaceutical company developing next-generation products to address the growing needs in oncology, is pleased to announce that it has successfully treated its first participant in the diagnostic US-based 64Cu SAR-bisPSMA trial for patients with biochemical recurrence (BCR) of prostate cancer.
COBRA (Copper-64 SAR-bisPSMA in Biochemically Recurrent prostAte cancer) is a Phase I/II Positron Emission Tomography (PET) trial of participants with BCR of prostate cancer following definitive therapy (NCT05249127[1]). It is a multi-centre, single arm, non-randomised, open-label trial of 64Cu-labelled SAR-bisPSMA in up to 50 participants. The primary objectives of the trial are to investigate safety and tolerability of 64Cu-SAR-bisPSMA as well as its ability to correctly detect recurrence of prostate cancer.
Clarity's Executive Chairman, Dr Alan Taylor, commented, "We are excited to have dosed the first participant in the COBRA trial at the Urology Cancer Center and GU Research Network (GURN) in Omaha, Nebraska, which continues to actively screen and recruit patients. We are very pleased to see our collaboration with Dr Luke Nordquist at GURN grow and evolve as we fully explore the many clinical and logistical benefits of Targeted Copper Theranostics (TCT)."
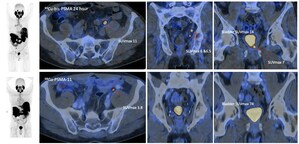
Prostate cancer is a key focus of Clarity's TCT program. Most recently, Clarity announced a collaboration with GURN on a diagnostic 64Cu SAR-bisPSMA investigator-initiated trial (IIT), X-Calibur (NCT05286840)[2], sponsored by Dr Luke Nordquist. The US-based theranostic 64Cu/67Cu SAR-bisPSMA trial, SECuRE (NCT04868604)[3], has been able to successfully image patients with metastatic castrate resistant prostate cancer from 1 hour to 72 hours post-injection. The diagnostic 64Cu SAR-bisPSMA trial in Australia, PROPELLER (NCT04839367)[4], is well underway, and will soon reach full recruitment in untreated, confirmed prostate cancer patients (i.e. pre-radical prostatectomy). Clarity has previously received advice from the FDA that its prostate diagnostic clinical program with 64Cu SAR-bisPSMA is addressing the two relevant patient populations for registration: pre-prostatectomy/pre-definitive treatment as well as patients with suspected biochemical recurrence.
Dr Luke Nordquist, CEO and Urologic Medical Oncologist at the Urology Cancer Center and GU Research Network in Omaha, Nebraska, commented, "We are very excited to have treated the first participant in the COBRA trial and look forward to continuing recruitment at GURN as the more hands-on experience with the TCT platform we gain, the more impressed with these next-generation theranostics we are. Apart from the favourable clinical data acquired in the SECuRE trial at GURN to date, which includes the ability to image tumours between 1 and 72 hours, SAR-bisPSMA enables us to address the significant backlog of patients who cannot access sufficient quantities of PSMA imaging agents based on gallium-68 (Ga-68) or fluorine-18 (F-18) due to the logistical issues of short half-life isotopes. The properties, including the longer half-life of Cu-64, may offer improvements in imaging disease and facilitate central manufacture of the diagnostics, meaning that we can provide critical imaging on-demand and in large scale, delivering the next-generation of technologies to prostate cancer patients and ensuring a timely and accurate diagnosis."
Dr Taylor said, "Treating the first patient in the COBRA trial is an important step in our prostate cancer program and we look forward to receiving preliminary results in participants with suspected recurrence of prostate cancer. We hope that SAR-bisPSMA will enable improved prostate cancer detection, including low volume disease, which is particularly important in this patient population where early and accurate diagnosis has significant implications for the patients' treatment outcome and prognosis. The preliminary data received from the PROPELLER and SECuRE trials to date is excellent as we have seen high uptake in tumours, and combined with centralised manufacture with on-demand delivery to any zip code in the continental US, this makes SAR-bisPSMA an ideal agent for the pursuit of our ultimate goal of improving treatment outcomes for cancer patients."
This announcement has been authorised for release by the Executive Chairman.
About Clarity Pharmaceuticals
Clarity is a clinical stage radiopharmaceutical company focused on the treatment of serious disease. The Company is a leader in innovative radiopharmaceuticals, developing targeted copper theranostics based on its SAR Technology Platform for the treatment of cancer in children and adults.
www.claritypharmaceuticals.com
About SAR-bisPSMA
SAR-bisPSMA derives its name from the word "bis", which reflects a novel approach of connecting two prostate-specific membrane antigen (PSMA) binding motifs to Clarity's proprietary sarcophagene (SAR) technology that securely holds copper isotopes inside a cage-like structure, called a chelator. Unlike other commercially available chelators, the SAR technology prevents copper leakage into the body. SAR-bisPSMA is a Targeted Copper Theranostic (TCT) that can be used with isotopes of copper-64 (Cu-64 or 64Cu) for imaging and copper-67 (Cu-67 or 67Cu for therapy).
About Prostate Cancer
Prostate cancer is the second most common cancer diagnosed in men globally and the fifth leading cause of cancer death worldwide[5]. The National Cancer Institute estimates in 2022 there will be 268,490 new cases of prostate cancer in the US and around 34,500 deaths from the disease[6].
References
1. ClinicalTrials.gov Identifier: NCT05249127 <https://clinicaltrials.gov/ct2/show/NCT05249127>
2. ClinicalTrials.gov Identifier: NCT05286840 <https://clinicaltrials.gov/ct2/show/NCT05286840>
3. ClinicalTrials.gov Identifier: NCT04868604 <https://clinicaltrials.gov/ct2/show/NCT04868604>
4. ClinicalTrials.gov Identifier: NCT04839367 <https://clinicaltrials.gov/ct2/show/NCT04839367>
5. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries <https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21660>
6. American Cancer Society, Cancer Statistics Center, <https://cancerstatisticscenter.cancer.org/?_ga=2.79808020.284532473.1620009137-1916069442.1615761164#!/cancer-site/Prostate>
SOURCE Clarity Pharmaceuticals







Share this article