
Cepheid Receives Emergency Use Authorization for Xpert Xpress CoV-2/Flu/RSV plus
New "plus" version of test adds a 3rd gene target to enhance detection of future SARS-CoV-2 variants
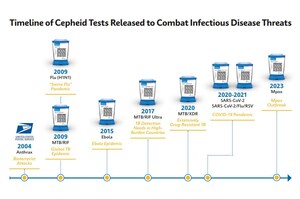
SUNNYVALE, Calif., Sept. 14, 2021 /PRNewswire/ -- Cepheid today announced it has received Emergency Use Authorization (EUA) from the U.S. Food & Drug Administration (FDA) for Xpert Xpress CoV-2/Flu/RSV plus, a rapid molecular diagnostic test for qualitative detection of the viruses causing COVID-19, Flu A, Flu B, and respiratory syncytial virus (RSV) infections from a single patient sample. The new plus version of the test provides a third gene target for SARS-CoV-2 detection to meet the challenge of future viral mutations. Xpert Xpress CoV-2/Flu/RSV plus is designed for use on any of Cepheid's over 35,000 GeneXpert® Systems placed worldwide, with results delivered in approximately 36 minutes.
Multiple variants of the virus that causes COVID-19 have been documented globally during the pandemic. Viruses constantly change through mutation, and new variants of a virus are expected to occur over time. Adding a 3rd gene target to the plus version of Cepheid's CoV-2/Flu/RSV test delivers broader coverage to mitigate the possible effects of future viral genetic drift.
"This respiratory season, healthcare providers may encounter a range of viral infections with symptoms that overlap with COVID-19, including Flu A, Flu B, and respiratory syncytial virus. Having a fast and accurate test that is designed to detect current and future variants of the viruses that cause COVID-19 and influenza will become increasingly important," said Dr. David Persing, MD, Ph.D., Chief Medical and Technology Officer at Cepheid. "The ability to collect one sample and run a single, highly-sensitive multiplexed test that detects and differentiates all four viruses will provide actionable results to inform better front-line decisions within our healthcare systems."
Xpert Xpress CoV-2/Flu/RSV plus is expected to begin shipping to US customers in the next few weeks.
Visit www.cepheid.com for more information, videos, and package inserts.
About Cepheid
Based in Sunnyvale, Calif., Cepheid is a leading molecular diagnostics company. Cepheid is dedicated to improving healthcare by developing, manufacturing, and marketing accurate yet easy-to-use molecular systems and tests. By automating highly complex and time-consuming manual procedures, the company's solutions deliver a better way for institutions of any size to perform sophisticated molecular diagnostic testing for organisms and genetic-based diseases. Through its strong molecular biology capabilities, the company is focusing on those applications where accurate, rapid, and actionable test results are needed most, such as managing infectious diseases and cancer. For more information, visit http://www.cepheid.com.
About Emergency Use Authorization Status
This test has not been FDA cleared or approved. It has been authorized by the FDA under an EUA for use by authorized laboratories. The test has been authorized only for the simultaneous qualitative detection and differentiation of nucleic acid from SARS-CoV-2, influenza A virus, influenza B virus, and RSV and not for any other viruses or pathogens. The test is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostic tests for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
For Cepheid Media Inquiries:
Darwa Peterson
[email protected]
SOURCE Cepheid







Share this article