
Centinel Spine Wins 2020 Spine Technology Award for its Two-level Indication for the prodisc® L Total Disc Replacement System
- Orthopedics this Week Spine Technology Award presented to Centinel Spine for its two-level indication approval by the FDA for the prodisc® L Total Disc Replacement (TDR) system
- Centinel Spine was selected for the award amid a record year of submissions and winning technologies
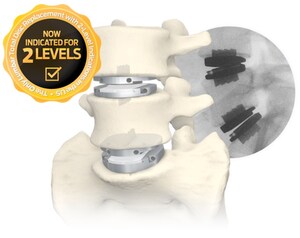
- prodisc L is the only total disc replacement system in the U.S. approved for two-level use in the lumbar spine
- Centinel Spine is the only company with FDA-approved cervical and lumbar total disc replacement devices
WEST CHESTER, Pa., Nov. 19, 2020 /PRNewswire/ -- Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced it has won a 2020 Orthopedics this Week Spine Technology Award for its two-level indication approval by the FDA for the prodisc® L Total Disc Replacement (TDR) system. The 12th Annual Spine Technology Awards recognize exemplary and innovative spine surgery technologies developed to improve spine care. Centinel Spine was presented and accepted the award today during a virtual Spine Technology Awards ceremony gala event organized by Orthopedics this Week.
According to Robin Young, founder and publisher of Orthopedics This Week, "2020 was a record year for submissions, and the winning technologies, as selected by a blue-ribbon panel of spine and neuro thought-leaders, are extraordinary. Centinel Spine's winning submission, for FDA approval for two-level indications for the prodisc L Lumbar Total Disc Replacement (TDR) system, is outstanding in that it is the only FDA-approved lumbar TDR device in the world that has been clinically-reviewed and found safe and effective for two-level use," Young concluded.
Centinel Spine's CEO, Steve Murray, stated, "I am honored to accept this award on behalf of Centinel Spine. As a company, we care deeply about advancing patient care and are proud to bring innovative technologies and expanded indications for use such as this to surgeons and patients. Centinel Spine will continue to advance spine care by bringing clinically-proven technologies to market that meet surgeon requirements and improve patient care," Murray concluded.
Centinel Spine announced FDA approval of two-level indications for the prodisc L system on April 14, 2020. The prodisc L device was first implanted in 2002 in the United States during concurrent IDE studies to obtain FDA approval for one- and two-level use. Results from the study have been published in numerous papers and are part of the over 540 published studies on the prodisc technology platform.
Centinel Spine stands alone as the only company with FDA-approved cervical and lumbar total disc replacement devices. The prodisc family of devices now consists of six devices, including an anterior and anterior-lateral approach lumbar disc replacement and four cervical disc replacement implants with a variety of endplate configurations designed to enable surgeons to better suit patient anatomy. New developments to the family of products include a recently-initiated two-level clinical trial comparing two new prodisc C device configurations with an approved TDR product as a control.
About Centinel Spine, LLC
Centinel Spine®, LLC is the largest privately-held spine company focused on anterior column reconstruction. The company offers a continuum of trusted, brand-name, motion-preserving and fusion solutions backed by over 30 years of clinical success—providing the most robust and clinically-proven technology platforms in the world for total disc replacement (prodisc®) and Integrated Interbody™ fusion (STALIF®).
Today, Centinel Spine advances its pioneering culture and continues its corporate mission to become the worldwide leading company addressing spinal disease anteriorly with the widest breadth & depth of technology platforms. Centinel Spine stands alone as the only company with comprehensive motion-preserving and fusion solutions for both cervical and lumbar anterior column reconstruction.
In June, 2019, the company entered into partnership with professional athlete Tiger Woods. Woods underwent spinal fusion surgery using Centinel Spine's STALIF M-Ti™ Anterior Lumbar Integrated Interbody fusion product in April 2017 to alleviate ongoing, debilitating pain in his back and legs.
Centinel Spine derived its name from the "Sentinel Sign", the radiographic confirmation of a successful fusion anterior to the interbody device.
For more information, please visit the company's website at www.CentinelSpine.com or contact:
Varun Gandhi
Chief Financial Officer
900 Airport Road, Suite 3B
West Chester, PA 19380
Phone: 484-887-8871
Email: [email protected]
SOURCE Centinel Spine, LLC






Share this article