
SHANGHAI, April 20, 2023 /PRNewswire/ -- CARsgen Therapeutics Holdings Limited (Stock Code: 2171.HK), a company focused on innovative CAR T-cell therapies for the treatment of hematologic malignancies and solid tumors, announced that CT041, an autologous CAR T-cell product candidate against the protein Claudin18.2 (CLDN18.2), has achieved IND clearance from the National Medical Products Administration (NMPA) for the postoperative adjuvant therapy of CLDN18.2 positive pancreatic cancer (PC).
Dr Raffaele Baffa, Chief Medical Officer of CARsgen, commented that "We are glad to receive the IND clearance from NMPA for the adjuvant treatment of CLDN18.2 positive pancreatic cancer using CT041. Pancreatic cancer remains a deadly disease with a dismal prognosis and lack of effective therapies. As indicated in previous clinical trials, CT041 has shown promising efficacies in treating pancreatic cancer. With this IND clearance, we look forward to exploring the use of CT041 for the adjuvant treatment and its potential to create deeper responses for pancreatic cancer patients. Driven by the vision of 'Making Cancer Curable', we will continue to explore the use of our CAR T-cells in the earlier line treatment of various cancer types."
About CT041
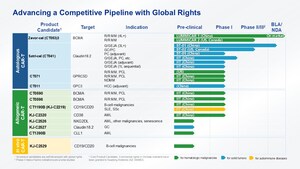
CT041 is an autologous CAR T-cell product candidate against the protein CLDN18.2 that has the potential to be the first-in-class globally. CT041 targets the treatment of CLDN18.2 positive solid tumors with a primary focus on gastric cancer/gastroesophageal junction cancer (GC/GEJ) and PC.
Active trials in CARsgen include investigator-initiated trials, a Phase Ib clinical trial for advanced GC/GEJ and PC and a confirmatory Phase II clinical trial for advanced GC/GEJ in China (CT041-ST-01, NCT04581473), and a Phase 1b/2 clinical trial for advanced gastric or pancreatic adenocarcinoma in North America (CT041-ST-02, NCT04404595). In January 2022, CT041 was granted Regenerative Medicine Advanced Therapy (RMAT) Designation by U.S. FDA for the treatment of advanced gastric or gastroesophageal junction adenocarcinoma with CLDN18.2 positive tumors. In November 2021, CT041 was granted PRIME Eligibility by the EMA for the treatment of advanced gastric cancer. In 2020 and 2021, CT041 received Orphan Drug designation from the U.S. FDA for the treatment of GC/GEJ and Orphan Medicinal Product designation from the EMA for the treatment of advanced gastric cancer. A Phase 2 clinical trial of CT041 in the U.S. is planned to initiate in the first half of 2023.
About CARsgen Therapeutics Holdings Limited
CARsgen is a biopharmaceutical company with operations in China and the U.S. and is focused on innovative CAR T-cell therapies for the treatment of hematologic malignancies and solid tumors. CARsgen has built an integrated cell therapy platform with in-house capabilities that span target discovery, antibody development, clinical trials, and commercial-scale manufacturing. CARsgen has internally developed novel technologies and a product pipeline with global rights to address major challenges of CAR T-cell therapies, such as improving the safety profile, enhancing the efficacy in treating solid tumors, and reducing treatment costs. CARsgen's vision is to become a global biopharmaceutical leader that brings innovative and differentiated cell therapies to cancer patients worldwide and makes cancer curable.
Contact Us
For more information, please visit https://www.carsgen.com/
SOURCE CARsgen Therapeutics






Share this article